Communication to Physicians
of
Diverse Patients
We appreciate this opportunity to share with you a model for achieving Therapeutic Equity for our American patients.
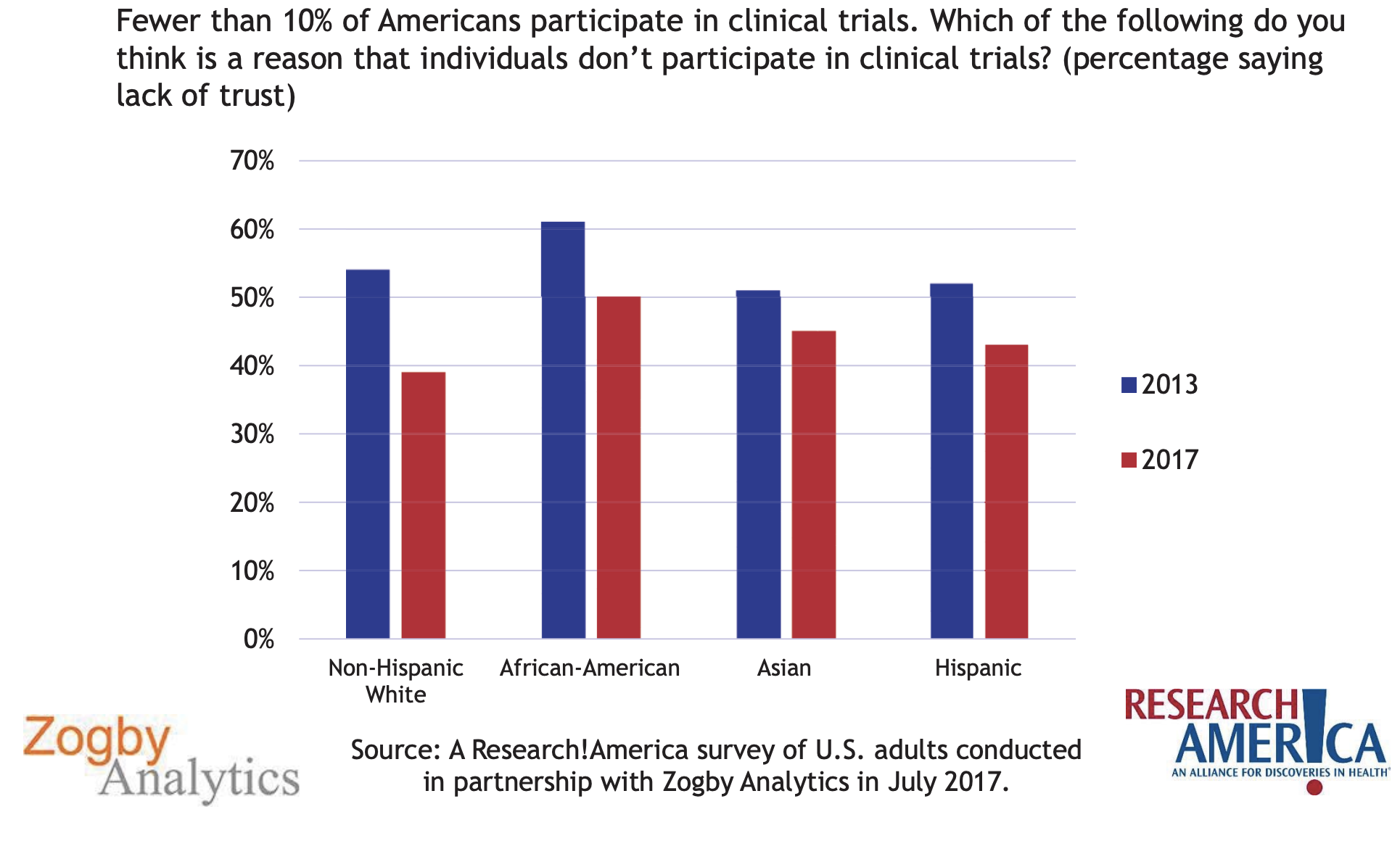
Routinely, the racial and ethnic diversity of American patients who are burdened by disease is poorly represented in the clinical trials data that support drug approvals and inform caregivers for the safe and effective use of drug treatments. The consequences of this data shortfall are rarely studied but can be dire. Consequences include ineffective treatment for some patients, prolonged or repeat hospitalizations, and delayed recognition of adverse events that can perpetuate health disparities and negatively impact the quality of patient care. Patients, however, have already spoken on this issue. Surveys indicate most of them recognize the value of clinical trials and the benefits of participation.
Zogby Analytics 2017
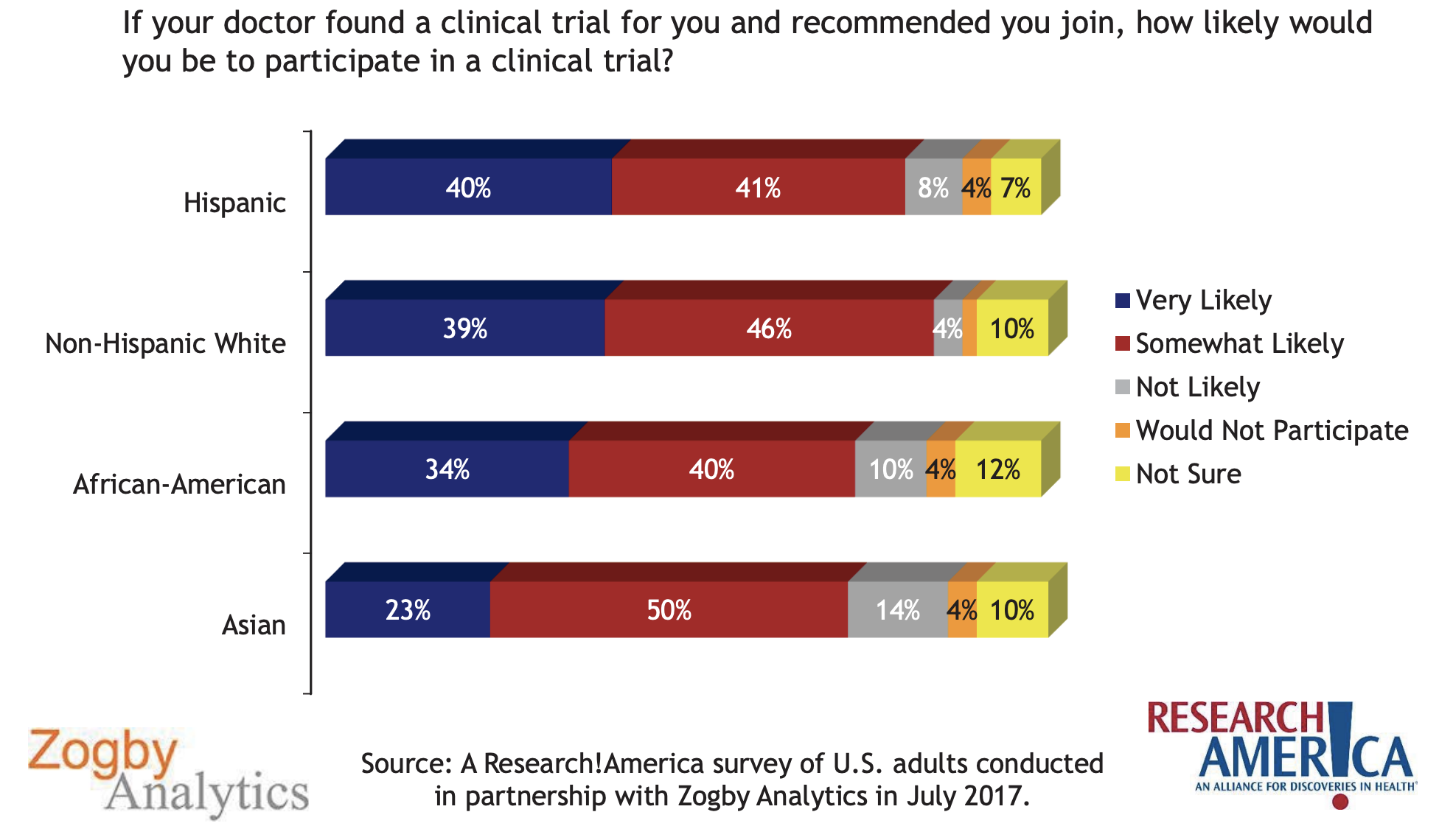
While mistrust is always an important issue, the data indicate mistrust is not insurmountable. Furthermore, patients identify their physicians as the preferred conduits through which they receive trustworthy information that can influence their willingness to participate in clinical trials.
Zogby Analytics 2017
Nearly 80% of the new drugs approved annually report inadequate data to prove safety and effectiveness in nonwhite patients. It may appear appropriate to assign the responsibility to regulators, policymakers, or global biopharmaceutical industry sponsors for this failure of the clinical evaluation of new medicines to routinely reflect the needs of a diverse American population. However, an important part of the solution may rest not with these entities, but in the trust relationship that exists between patients, their physician-caregivers and the health institutions that often employ them. This trust relationships carries with it significant responsibility for establishing and supporting pathways by which physicians and patients gain the essential knowledge and tools that allow them both to exert their best desired influence over the clinical trials processes that impact their daily lives. This requires that physicians gain the knowledge, understanding and current information to share with their patients with minimal disruption to patient care activities.
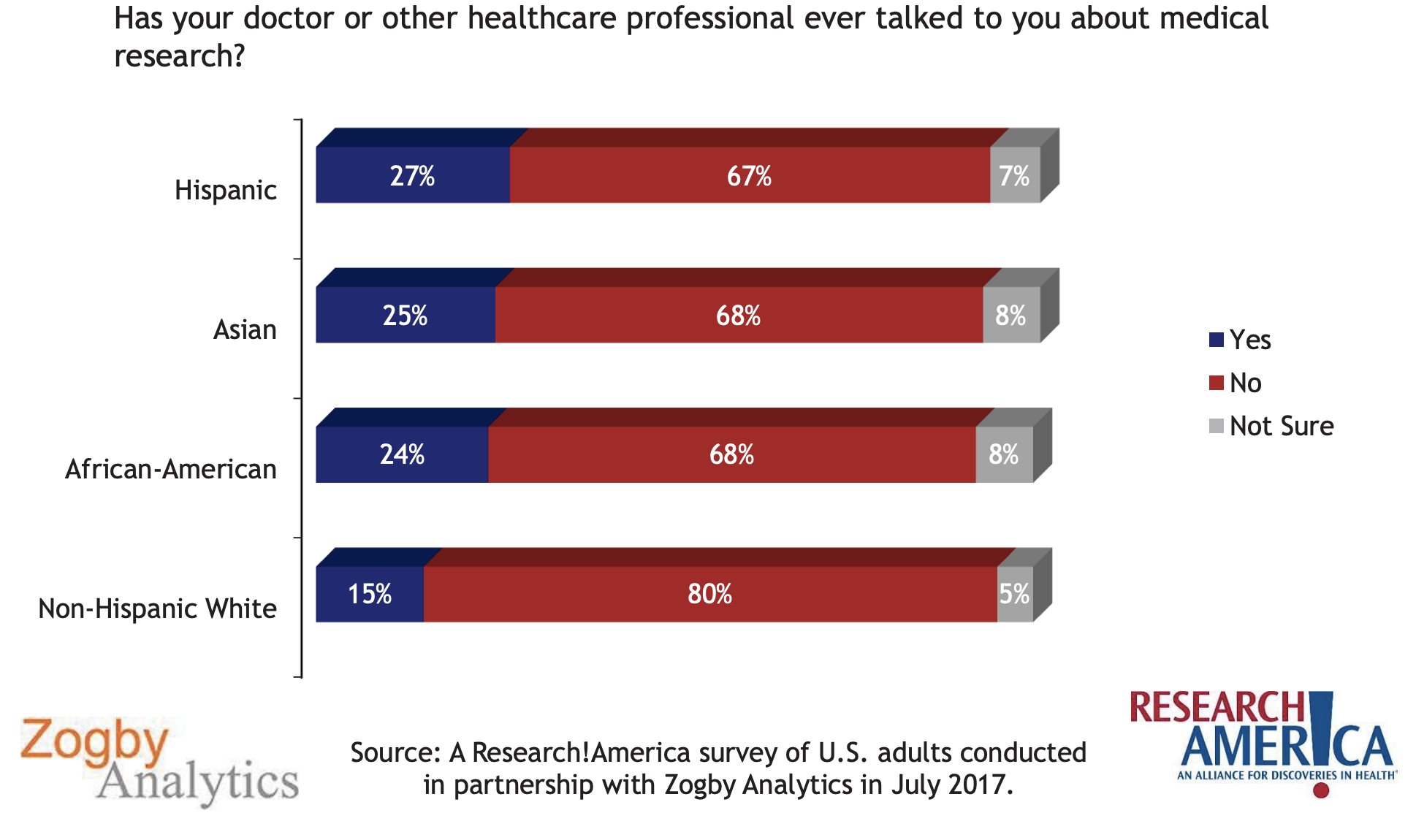
All medical professionals have the mission to deliver the best quality of patient care they can provide and to “do no harm.” Therefore, all medical professionals who have earned the trust of patients should recognize their responsibility for knowing enough about clinical research to adequately inform their patients. Unfortunately, lack of involvement by many physicians and affiliated institutions inevitably leads to lack of involvement for thousands of underrepresented patients who trust them. This circumstance helps to sustain an unintentional barrier to patient knowledge of trials and limits their well-informed participation with the potential harm of perpetuating the status quo of health disparities.
Zogby Analytics 2017
In the effort to prepare every trusted physician to be an informed resource for patients on clinical research participation, the rapidly changing environment of physicians as employees must be addressed. Over 70% of physicians practicing in the U.S. now do so as employees of a health institution or other corporate entity. Practicing in this capacity often limits the physician’s ability to make the decision and/or perform the functions required to participate in clinical trials. Physicians and their institutions may be inhibited by the traditional barriers of concerns for loss of patients to clinical trial referrals, concerns about loss of patient trust, lack of recognition of the consequences of underrepresentation, concerns for workflow disruptions, as well as concerns about actual or apparent conflicts of interest. These same physicians in service to a diverse population will often identify the elimination of health disparities as important to their mission. However, they may be unaware of their role as a barrier to clinical trial participation by the many patients who trust them.
The clinical research industry invests many millions of dollars in efforts to connect with patients about clinical trials. We believe greater benefit can be achieved from educating and informing of trusted physicians about clinical trials and better integrating them into the process. This requires processes/tools that avoid significant practice workflow disruptions and avoiding the perception of inappropriate influences and conflicts of interest. Our solution is a sustainable, HIPAA compliant and transparent process that addresses these challenges and establishes benefits beyond clinical trials. It includes a unique, on-demand training platform accessible via web portal and designed to equip physicians with the knowledge of clinical drug research necessary to respond to patient questions and, if interested, to become successful investigators. It does not support “pay for patient referrals,” but compensates physicians for investing the time and effort in training and reviewing clinical trial information. The platform also assists in sharing information from physicians to eligible patients about clinical trials and empowering them to make their own decisions regarding participation.
We seek the training and participation of physicians who share the vision for helping their patients by eliminating health disparities attributable to inadequate clinical trials data. We seek collaboration with those who are willing to work with us to establish the mutually beneficial model for the pursuit of Therapeutic Equity. Beyond the significant clinical research impact, our model claims utility in educating patients, nurturing the physician-patient trust relationship, and empowering physician and patient influence on clinical research as well as empowering patient self-care. We welcome you to join with us in expediting the integration of inclusive, efficient clinical research into better patient care.
Send a message to connect with me or my staff here.
James H. Powell, MD
Chief Medical Officer, knowRX, Inc.

